New Compound to Double Effectiveness of Redox Flow Battery
Growing global energy demands drive the need for technologies that can address energy storage at the grid and microgrid scale, thereby enabling the incorporation of distributed renewable resources such as solar and wind. Redox flow batteries (RFBs) are suitable as they decouple power and energy densities for straightforward scaling based on electrode stack size and reservoir volume.
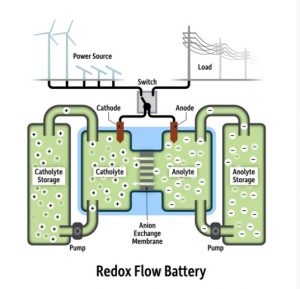
Image: University of Rochester/Michael Osadciw
Unlike secondary batteries that contain solid-phase electrolytes and migratory ions as charge carriers, RFBs take advantage of electrolyte solutions typically consisting of a solvent, a supporting electrolyte, and an electroactive species that can cycle in its redox states.
Viable devices have focused on aqueous electrolytes to date, which use inorganic salts as charge carriers.
Efficiency: At par with the Li-ion applications
As storage grows into an ever more important part of the energy system, redox flow batteries are often touted as a technology that could rival lithium-ion applications in the large-scale sector.
Economical alternatives
Finding affordable chemicals that can carry sufficient charge for long periods without degrading, however, remains a challenge to the technology’s widespread adoption – although some commercial projects utilizing redox flow batteries are already underway.
Latest research
Scientists from University of Rochester, working jointly with colleagues from University of Buffalo, have discovered a promising compound for use in redox flow batteries, which they say could transform the energy storage landscape.
How did they do it?

Chemical Science, The Royal Society of Chemistry
Rochester and Buffalo researchers used a metal-oxide cluster discovered originally by German chemist Johann Spandl, finding that it could be store charge in a redox flow battery. Stability was the only hindrance!
By undertaking a ‘simple molecular modification’, however, the researchers claimed to increase the stability of the material and double the amount of energy that could be stored in same-size battery.
“Energy storage applications with polyoxometalates are pretty rare in the literature,” lead researcher Lauren VanGelder told The TrickyScribe. “There are maybe one or two examples prior to ours, and they didn’t really maximize the potential of these systems.”
The breakthrough!
Key to the compound’s potential is the use of readily available materials: “What’s really cool about this work is the way we can generate the ethoxide and methoxide clusters by using methanol and ethanol.
Both of these reagents are inexpensive, readily available and safe to use. The metal and oxygen atoms that compose the remainder of the cluster are earth-abundant elements.
The straightforward, efficient synthesis of this system is a totally new direction in charge-carrier development that, we believe, will set a new standard in the field.”
The two universities have now applied for a grant from the National Science Foundation to further refine the materials for use in commercial redox flow batteries.
Backgrounder
It may be mentioned here that non-aqueous redox flow batteries have emerged as promising systems for large-capacity, reversible energy storage, capable of meeting the variable demands of the electrical grid.
Here, we investigate the potential for a series of Lindqvist polyoxovanadate-alkoxide (POV-alkoxide) clusters, [V6O7(OR)12] (R =CH3, C2H5), to serve as the electroactive species for symmetric, non-aqueous redox flow battery.
We demonstrate that the physical and electrochemical properties of these POV-alkoxides make them suitable for applications in redox flow batteries, as well as the ability for ligand modification at the bridging alkoxide moieties to yield significant improvements in cluster stability during charge-discharge cycling.
Metal–oxide core remains intact upon deep charge–discharge cycling, enabling extremely high coulombic efficiencies (somewhere around 97%) with minimal overpotential losses (close to 0.3 V). Furthermore, the bulky POV-alkoxide demonstrates significant resistance to deleterious crossover, which will lead to improved lifetime and efficiency in a redox flow battery.
Total Views: 2,93,295



